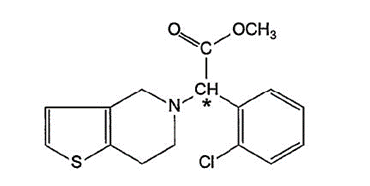
Clopidogrel is the common name of the dextrorotatory isomer of the chemical compound named methyl alpha-5(4,5,6,7-tetrahydro(3,2-c)thienopyridyl)(2-chlorophenyl) -acetate. The product has the property of inhibiting the aggregation of blood platelets, and is used to treat or prevent blood-thrombotic events such as heart attacks and strokes. Claim 3 of the patent is for:
Hydrogen sulfate of the dextro-rotatory isomer of methyl alpha-5(4,5,6,7-tetrahydro(3,2-c)thienopyridyl)(2-chlorophenyl)-acetate substantially separated from the levo-rotatory isomer.
Sanofi sued after Apotex filed an Abbreviated New Drug Application (ANDA) for FDA approval to sell clopidogrel bisulfate, along with a paragraph IV certification that the ’265 patent is invalid. A proposed settlement fell apart, the statutory stay expired, the FDA approved the Apotex ANDA, and Apotex starting selling its generic clopidogrel bisulfate product.
Sanofi then moved for a preliminary injunction, noting that infringement was conceded by Apotex. The district court found that Sanofi was likely to succeed on the merits of the validity and enforceability of the ’265 patent, granting the injunction. The Court of Appeals for the Federal Circuit affirmed the district court’s rulings and sent it back so that the district court could review all aspects at trial and the district court ruled that the ’265 patent is valid and enforceable.
This appeal is focused on the question of patentability of this dextrorotatory isomer in view of its known racemate described in earlier Sanofi patents, specifically, Sanofi’s US Pat. No. 4,529,596 and Canadian Pat. No. 1,194,875. Both reference patents are derived from the same French priority filing and are prior art against the ’265 patent.
Sanofi discovered that certain compounds of the class known as thienopyridines (compounds having a thiene ring fused to a pyridine ring) have the property of inhibiting blood platelet aggregation. Sanofi initially synthesized and evaluated several hundred chemical modifications and derivatives of thienopyridines, seeking optimum anti-platelet aggregation properties with minimal undesirable effects.
They eventually selected for development the compound “ticlopidine.” After development, ticlopidine was approved in the US for use as an anti-thrombotic agent. This was approved but had possible adverse effects of rarely occurring but serious blood disorders, neutropenia and thrombotic thrombocytopenic purpura, associated with prolonged usage of ticlopidine.
Sanofi then synthesized and evaluated several hundred additional thienopyridine derivatives, including a class of compounds having the following general structure, wherein one of the hydrogen atoms on the bridge carbon atom (marked with an asterisk), is replaced with an ester, carboxylic acid, or amide group. This class is the subject of the ’596 patent (and the counterpart Canadian ’875 patent), and had good anti–platelet aggregation properties.
Focusing on the ’596 patent, the specification includes twenty-one examples of specific compounds, including a compound designated as PCR 4099. This compound has the chemical name methyl alpha-5(4,5,6,7-tetrahydro(3,2-c)thieno pyridyl)(2-chlorophenyl)-acetate, with the acronym MATTPCA. PCR 4099 as the hydrochloride salt was selected for commercial development as a potential replacement for ticlopidine in light of its improved platelet inhibition and toxicity profile.
However, PCR 4099 still raised toxicity concerns, for at very high doses it caused convulsions in laboratory animals. Thus the research efforts continued, concurrently with the clinical and commercial development of PCR 4099. Sanofi states that about 1500 compounds in this general class were synthesized, of which about 600, including PCR 4099, were chiral thienopyridines.
Enantiomers are spatial isomers, also called stereoisomers, identified and distinguished by their optical characteristics when a purified solution of the separated isomers is exposed to plane-polarized light. Enantiomers generally are formed in equal amounts, to produce what is called a racemate; the racemate is optically neutral.
Believing that there was no advantage to separation of the enantiomers of thienopyridines, and no other racemates were separated until November 1985 when Sanofi then determined the biological properties of the enantiomers of PCR 4099, and found that they had the rare characteristic of “absolute stereoselectivity”: the dextrorotatory enantiomer provided all of the favorable antiplatelet activity but with no significant neurotoxicity, while the levorotatory enantiomer produced no antiplatelet activity but virtually all of the neurotoxicity. This kind of stereoselectivity is rare.
Sanofi filed a patent application directed to clopidogrel and certain salts and pharmaceutical compositions. The ’265 specification explains that the racemate of the same chemical formula was described in the earlier French ’247 patent, which corresponds to the earlier U.S. ’596 patent. The ’265 patent discusses the unusual stereoselectivity of the biological properties as between the dextrorotatory and the levorotatory enantiomers. The US patent examiner, who had also examined the ’596 patent, allowed the claims after requiring that the ’265 claims make clear that the dextro- and levo- enantiomers are “substantially separated.”
Apotex appealed on the issues of anticipation and obviousness.
Claimed subject matter is “anticipated” when it is not new; that is, when it was previously known. Invalidation on this ground requires that every element and limitation of the claim was previously described in a single prior art reference, either expressly or inherently, so as to place a person of ordinary skill in possession of the invention.
Apotex argued that the reference shows the specific racemate PCR 4099 and states that the compounds in the reference have enantiomers and that the enantiomers are included in the invention. Apotex claimed that the properties of the enantiomers of PCR 4099 are inherently and necessarily present in its known racemate, such that when the enantiomers are separated the previously observed properties are “immediately recognized” in one or the other enantiomer.
The question hinged on whether a generic disclosure necessarily anticipates everything within the genus, and recognized that the answer depends on the factual aspects of the specific disclosure and the particular products at issue. In this case the district court said the references’ general statements that these compounds consist of enantiomers didn’t make an anticipating disclosure of the separated dextrorotatory enantiomer of PCR 4099.
Apotex also tried arguing that although the reference did not state that the disclosed compound was a racemate, it would have been known to one of ordinary skill that synthetically produced chiral compounds are racemic.
This was knocked down:
The district court did not clearly err in finding that the statements in the ’596 patent and its Canadian counterpart that the products therein consist of enantiomers are not a description of the specific dextrorotatory enantiomer clopidogrel or a suggestion of its unusual stereospecific properties. The knowledge that enantiomers may be separated is not “anticipation” of a specific enantiomer that has not been separated, identified, and characterized. The district court correctly held that neither the ’596 patent nor its Canadian counterpart contains an anticipating disclosure of the subject matter of claim 3 of the ’265 patent.
The court also looked at the question of obviousness. For chemical compounds, the structure of the compound and its properties are inseparable considerations in the obviousness determination. Previous case law established that when dealing with analytical procedures where there is a close structural similarity between a new chemical compound and prior art compounds, it is generally deemed to create a prima facie case of obviousness, shifting to the patentee the burden of coming forward with evidence of nonobviousness.
Apotex argued that the recognition in the prior art that PCR 4099 is composed of enantiomers outweighs the effect of any unexpected or unpredictable properties of the separated dextrorotatory enantiomer.
The court also shot this down:
The determination of obviousness is dependent on the facts of each case. See Graham, 383 U.S. at 17-18. In Forest Laboratories, 501 F.3d at 1269, this court affirmed that the (+) enantiomer of citalopram would not have been obvious in light of the known racemate, when it was shown that the therapeutic properties of the (+) enantiomer were unexpected. In contrast, in Aventis Pharma Deutschland GmbH v. Lupin, Ltd. 499 F.3d 1293, 1302 (Fed. Cir. 2007), this court held that the ramipril isomer’s potency was “precisely what one would expect, as compared to a mixture containing other, inert or near-inert stereoisomers.” Apotex argues that Aventis is the closer analogy, but the evidence was directly contrary to that position. The district court entered extensive findings in this case on the unexpected and unpredictable properties of clopidogrel, and there was no contrary evidence suggesting, based on the prior art, that the stereoselective properties were “precisely what one would expect,” as in Aventis.
Update: As noted in the comments, Teflon® is a brand of poly(tetrafluoroethylene) or poly(tetrafluoroethene) (PTFE), a synthetic fluoropolymer used as a non-stick coating for pans and other cookware, and is registered trademark of E. I. du Pont de Nemours and Company.

are there references to above data re number of molecules synthesized and tested, eg.,
Sanofi then synthesized and evaluated several hundred additional thienopyridine derivatives,
I am not a noun.
just quoting what was written, would still like to have more documentation of what was written re SAR, JOE